Research shows the path toward trachoma elimination
November / December 2012 | Volume 11, Issue 6
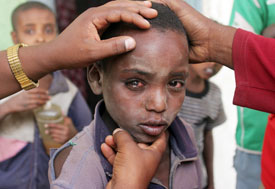
Photo by Raul Vasquez/ORBIS
The National Eye Institute at the NIH supports
research to determine the most effective ways
to deploy antibiotics to eliminate trachoma,
which currently affects about 40 million people,
mostly in the developing world.
Repeated mass treatments with antibiotics can greatly reduce the occurrence of trachoma, a bacterial eye infection that causes blindness in millions of people, NIH-supported research has shown. In addition to paving the way to eliminate the disease, which largely strikes developing countries, studies indicate the therapy also protects communities from other infections, reducing child deaths from pneumonia, diarrhea and malaria by 50 percent.
After years of targeted funding and research,
trachoma has been wiped out in some regions and the WHO aims to vanquish it in remaining places by 2020. Research funded by the NIH's
National Eye Institute (NEI) is playing a crucial role by identifying effective antibiotic regimens to combat the scourge. These include determining how often to administer treatment, what portion of a community must be reached and how to prevent re-infection.
The main treatment for active trachoma, caused by the
Chlamydia trachomatis bacterium, is the antibiotic azithromycin. Mass treatment became economically feasible only in 1998 when the International Trachoma Initiative began making substantial quantities of azithromycin available at no cost. "We now had a single-dose antibiotic, which provided the impetus to fight this disease," recalls Dr. Sheila K. West, NEI grantee and professor at Johns Hopkins University.
The idea of treating entire communities at once is similar to vaccination programs against diseases such as polio and typhoid. Both infected and non-infected individuals in a community are inoculated, to reduce the pool of infection and prevent the eruption of new cases. Unlike vaccinations, though, mass treatments with antibiotics typically need to be repeated numerous times.
Studying ways to prevent infection
This raised questions of how frequently and for how many years treatments should be administered to keep infection at bay. It might be impossible to reach every person in a community, so how widespread did the treatment need to be to produce "herd immunity," as in vaccination programs? Should only children receive the antibiotic or also adults? Would periodic mass treatments with antibiotics increase the risk of drug resistance?
NEI focused on these important issues, funding researchers to conduct several large community-based studies in different locations with widespread trachoma infection. Early results showed a single mass treatment with azithromycin reduced disease prevalence in a community, but infection returned within a year in the most severely affected areas. Another study tested the impact of multiple mass treatment rounds and found communities needed more than seven - and perhaps as many as 10 - annual mass treatments to conquer trachoma. Subsequent trials confirmed that long-term reduction and even elimination of the disease is possible with repeated mass antibiotic treatments involving most residents of a community over several years.
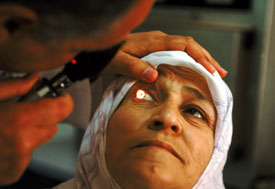
Photo by Alan Gignoux/World Bank
The National Eye Institute currently funds
more than two dozen grants in 15 non-U.S.
countries, supporting global health research
in genetic, infectious and environmental eye
diseases.
"By giving treatment on an annual basis, you eventually catch up and prevent the disease from flourishing," West said. "The question now is not so much how frequently you do it, but whether you can get away with treating just kids versus treating everybody."
Meanwhile in Ethiopia, NEI-funded researcher Dr. Thomas M. Lietman led a clinical trial involving 24 communities to determine whether mass treatment of children younger than 11 would protect the entire population. Working in collaboration with the Carter Center and the health ministry, Lietman discovered after one year that trachoma occurrences among children given azithromycin had plunged from 48 percent to less than 4 percent. Infection also dropped significantly in older, untreated children and adults, suggesting that repeatedly treating younger children might be a cost-effective strategy to control infection throughout a community.
Not only did the antibiotic ward off eye disease, but Lietman and his colleagues discovered another benefit - child deaths dropped by half in treated communities, suggesting that azithromycin helps mitigate potentially lethal infections such as pneumonia, diarrhea and malaria.
However, the potential for drug resistance to antibiotics is a key concern. Studies have shown the bacterium
Streptococcus pneumonia is capable of building resistance, but the risk diminishes several months after treatment. The long-term effect of administering multiple rounds of azithromycin is still unknown.

Photo by Kiran Ridley/ORBIS
Trachoma is the leading cause of preventable
blindness. It spreads through contact with hands,
clothing or flies.
Preventing blindness and re-infection
Repeated trachoma infections often lead to trichiasis, a condition of turned-in eyelashes that scratch the eyeballs to the point of blindness. In such cases, mass antibiotic treatment does not prevent vision loss, although surgery can. Yet, trichiasis recurs in half of post-surgical cases. To improve outcomes, the NEI funded the Surgery for Trichiasis, Antibiotics to Prevent Recurrence (STAR) trial. West and her colleagues in Ethiopia randomized one group of patients with trichiasis to receive azithromycin after surgery; the other group received standard postsurgical care, which involved topical tetracycline ointment. Azithromycin reduced overall trichiasis recurrence by one-third and severe trichiasis recurrence by almost half, compared with standard treatment.
NEI-funded research over the past decade has answered these and other scientific questions necessary to tackle trachoma, laying the groundwork for other organizations to become involved in elimination efforts. For example, the Bill and Melinda Gates Foundation has awarded $12 million to Johns Hopkins University to continue its work. With more than 40 million people suffering from trachoma throughout the developing world, elimination will be an enormous task, but the treatment and prevention approaches needed for success are now more clearly defined.
"NEI stepped up to the plate early on by funding trachoma research, hoping that would lead to other partners to come in and fill in the gaps, and that has now happened," West said. "The goal of elimination of blinding trachoma by 2020 will be a challenge. Research to inform programs on strategies to achieve that goal will be a key component to success."
More Information
Articles in the Focus on the National Eye Institute at NIH section were produced by Cathy Kristiansen, with contributions from Christopher G. Thomas, Tom Hoglund and Richard S. Fisher.
To view Adobe PDF files,
download current, free accessible plug-ins from Adobe's website.