Fogarty-supported research revolutionizes hydrocephalus care
September / October 2018 | Volume 17, Number 5
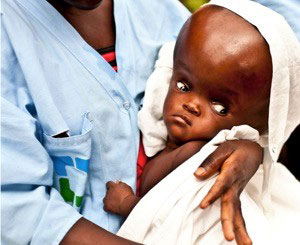
A health care worker holds a baby with hydrocephalus
at the CURE pediatric neurosurgery hospital in Uganda.
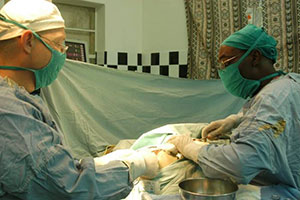
Photos courtesy of Dr. Benjamin Warf
Drs. Benjamin Warf (left) and John Mugamba operate
on a child with hydrocephalus at the CURE hospital
in Uganda.
By Karin Zeitvogel
A treatment pioneered and proven with Fogarty support has revolutionized care for hydrocephalus and saved money - and probably lives - in resource-poor and wealthy countries alike. Developed by Dr. Benjamin Warf after he noticed “an enormous number of babies with hydrocephalus” presenting at a Ugandan children's neurosurgery clinic, the treatment allows doctors to avoid placing a shunt in babies to drain excess cerebrospinal fluid from the brain.
“This is important because half of shunts fail within two years of placement and all fail within five to 10 years,” said Warf. “When they fail, the child has to have emergency surgery or they’ll die from elevated intracranial pressure. But in sub-Saharan Africa and other limited-resource regions, many patients don’t have access to emergency medical systems where they can get immediate care.”
Warf himself placed shunts, the predominant treatment worldwide for hydrocephalus for several decades, in hundreds of babies with hydrocephalus during his first few years in Uganda, but in the back of his mind was the nagging thought that, while he was treating one problem, he was creating another - shunt dependence. So he set about developing a novel treatment for hydrocephalus, combining two methods that had been used individually before, with limited success. One, endoscopic third ventriculostomy (ETV), involves making an opening in one of the ventricles, or cavities, of the brain to allow fluid to drain, and the other, choroid plexus cauterization (CPC), reduces the amount of cerebrospinal fluid the brain produces.
Scientists knew that ETV-CPC is less likely to fail over the long term but didn’t reduce the size of the ventricle as much as shunt placement did, raising concerns that cognitive development would be compromised in children who had the new procedure. So, with a grant through Fogarty’s brain disorders program, largely funded by the
Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), Warf and his colleagues ran a randomized controlled trial to compare outcomes after ETV-CPC and shunting in babies with post-infectious hydrocephalus.

© John D. and Catherine T.
MacArthur Foundation, used
with permission
Dr. Benjamin Warf
Twelve months after surgery, they assessed the neurological development of 100 infants enrolled in the study conducted at the CURE pediatric neurosurgery hospital in Mbale, Uganda. Their findings, published in the
New England Journal of Medicine, showed that children in the shunt group had smaller ventricles a year post-operation than the ETV-CPC group, but brain growth and developmental outcomes for the two groups were the same. For the first time, research was showing that developmental outcome correlated with brain volume, not ventricle size. “For the past 50 years, hydrocephalus treatment has been directed at making the ventricle smaller, but we found that what’s important is not ventricle size but whether the brain is growing or not,” said Warf.
The children will be followed for five years after surgery to allow Warf and his colleagues to continue to monitor and assess their development and brain growth.
Warf received a MacArthur Genius award in 2012 for developing ETV-CPC and advancing standards of, and access to, health care in both the developed and poorest regions of the world.
“ETV-CPC is an example of how helping people elsewhere also allows us to make gains for Americans,” he said. U.S. pediatric neurosurgeons have trained in the new technique in Uganda, where the large patient population allows them to perform multiple hydrocephalus operations a day. ETV-CPC has been taken up at major pediatric neurosurgery centers in North America, including Boston Children’s Hospital, where Warf has worked since he left Uganda in 2009. The number of shunts placed by pediatric neurosurgeons at Boston Children’s has been reduced by around a third, meaning fewer revisions when a shunt fails.
Estimates put the total cost in the U.S. of maintaining shunt function at between $1 billion and $2 billion a year, so placing fewer shunts is expected to lead to “big cost-savings and a decrease in morbidity and mortality from lifetime shunt dependence,” said Warf.
More Information
To view Adobe PDF files,
download current, free accessible plug-ins from Adobe's website.