Faster new test can prevent tuberculosis deaths
September / October 2012 | Volume 11, Issue 5
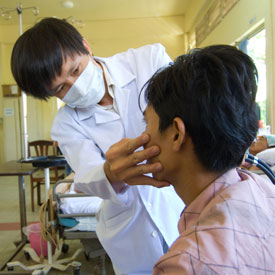
Photo by David Snyder for Fogarty/NIH
A fast, new diagnostic test, developed with NIH support,
will help prevent TB deaths.
The time it takes to diagnose a patient with tuberculosis (TB) is a matter of life and death in many parts of the world. Testing methods can take weeks and patients remain untreated and contagious in the meantime. But a new test developed with NIH support promises results in under two hours, so the patient can receive medicine the same day. It also reveals whether the patient harbors drug-resistant bacteria and needs special treatment.
TB is widespread, with a third of the world's population infected. Most people remain well, but those with weakened immune systems, such as HIV/AIDS patients or under-nourished children, can become seriously ill. In 2010, about 1.4 million people died of TB, primarily in developing countries.
Currently, health care workers detect TB by various methods, depending on resources, ranging from physical examinations to chest X-rays to blood or sputum tests. The NIH and the Bill and the Melinda Gates Foundation funded research to find a test that was quick, able to screen for multi-drug resistance and automated to limit human error. Rather than starting from scratch, the scientists adapted a test developed several years ago to detect anthrax sent through the U.S. Postal Service.
"We wanted a highly sensitive molecular test" that would be speedy, simple and detect multidrug-resistant TB, said Dr. Mark Perkins, chief scientific officer at the Foundation for Innovative New Designs (FIND), which is funded by Gates and produced the new TB test. Perkins, a former research fellow at the National Institute of Allergy and Infectious Diseases, spoke recently at the NIH.
Perkins and his team came up with Xpert MTB/RIF, an automated nucleic acid amplification test that comes in a self-contained, cartridge-based platform. From a sputum sample, it detects Mycobacterium tuberculosis and pinpoints if it has drug resistance, Perkins said. Resistance is particularly problematic because if it is undetected, patients receive inappropriate medicines.
The WHO endorsed the new test, predicting it would triple the diagnoses of resistant-TB and double the diagnosis of TB/HIV cases in high-rate areas. The test was initially priced at $18, but negotiations with manufacturers brought that down to $10, an amount that Perkins says he hope will drop further with higher-volume sales.
The lecture was sponsored by the Trans-NIH Global Health Working Group, formed to highlight opportunities for innovative research that will benefit low-resource countries.
More Information
To view Adobe PDF files,
download current, free accessible plug-ins from Adobe's website.