First-ever Alzheimer's prevention trial to take place in Colombia
May 15, 2012
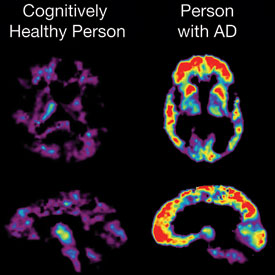
Image courtesy of the National Institute on Aging
The first-ever Alzheimer's prevention trial will investigate whether a drug can delay - or even stop - the disease in a group of Colombian subjects who are genetically destined to develop it.
In collaboration with the NIH, Banner Alzheimer's Institute, the University of Antioquia in Colombia and Genentech, announce the first‐ever prevention trial in cognitively healthy individuals who are destined to develop Alzheimer's disease because of their genetic history. This groundbreaking study - the first to investigate whether an anti-amyloid treatment can stave off the disease - will span two countries and help launch a new era of prevention research in the urgent fight against Alzheimer's.
The $100 million trial is the cornerstone of a new international collaborative, the Alzheimer's Prevention Initiative (API), formed to accelerate the evaluation of promising but unproven prevention therapies. It will study an experimental anti-amyloid antibody treatment called crenezumab in approximately 300 people from an extraordinarily large extended family in Colombia, who share a rare genetic mutation that typically triggers Alzheimer's symptoms around age 45. The trial will also include a smaller number of individuals in the United States. The API team will collaborate with researchers from the NIH-supported Dominantly Inherited Alzheimer's Network to identify and recruit the U.S. participants.
The trial is designed to determine whether the drug can reduce participants' chances of developing the disease's disabling and irreversible symptoms, preserve memory and thinking abilities, and slow the progression of Alzheimer's biomarkers.
The project is part of the U.S. national plan to address Alzheimer's Disease.
More Information
To view Adobe PDF files,
download current, free accessible plug-ins from Adobe's website.